You can’t beat the magic of Christmas. My kids love everything to do with the holiday, from Christmas decorations in the mall to delicious treats made at home. Of course, just because Christmas is around the corner doesn’t mean the learning can stop! That is why I love to give my kids educational activities with a holiday twist. I love these salt crystal ornaments because they are the perfect Christmas science experiment and you can use them as real tree ornaments!

* This post may contain affiliate links for your convenience. Click here for my full disclosure.
We prefer salt crystals above other types of homemade crystals because they are durable and just about no-fail. The square and rectangle crystals they make are pretty, too!
Salt Crystal Ornaments: Christmas Science Experiment

What you’ll need to make salt crystal ornaments:
- Chenille stems
- Salt
- Mason jars (1 for each ornament)
- Scissors
- Clothespins (1 for each ornament)

Before starting the experiment, shape your chenille stems into your ornament shapes. We chose to make a round ornament and a tree-shaped ornament, but you can make whatever shape you like!
We like to use chenille stems to suspend our shapes in the salt water because they are stiff enough to hold the ornament exactly where you want them without worrying about the ornament floating to the top of the jar and ruining the crystal design.
On the stove, heat 2 cups of water per mason jar until it boils. Add in salt until crystals form on the surface of the boiling water. For two mason jars, it will take about half of a regular-sized container of salt.

Use the clothespins and chenille stems to suspend the ornaments in the salt water solution.
Wait 24 hours.
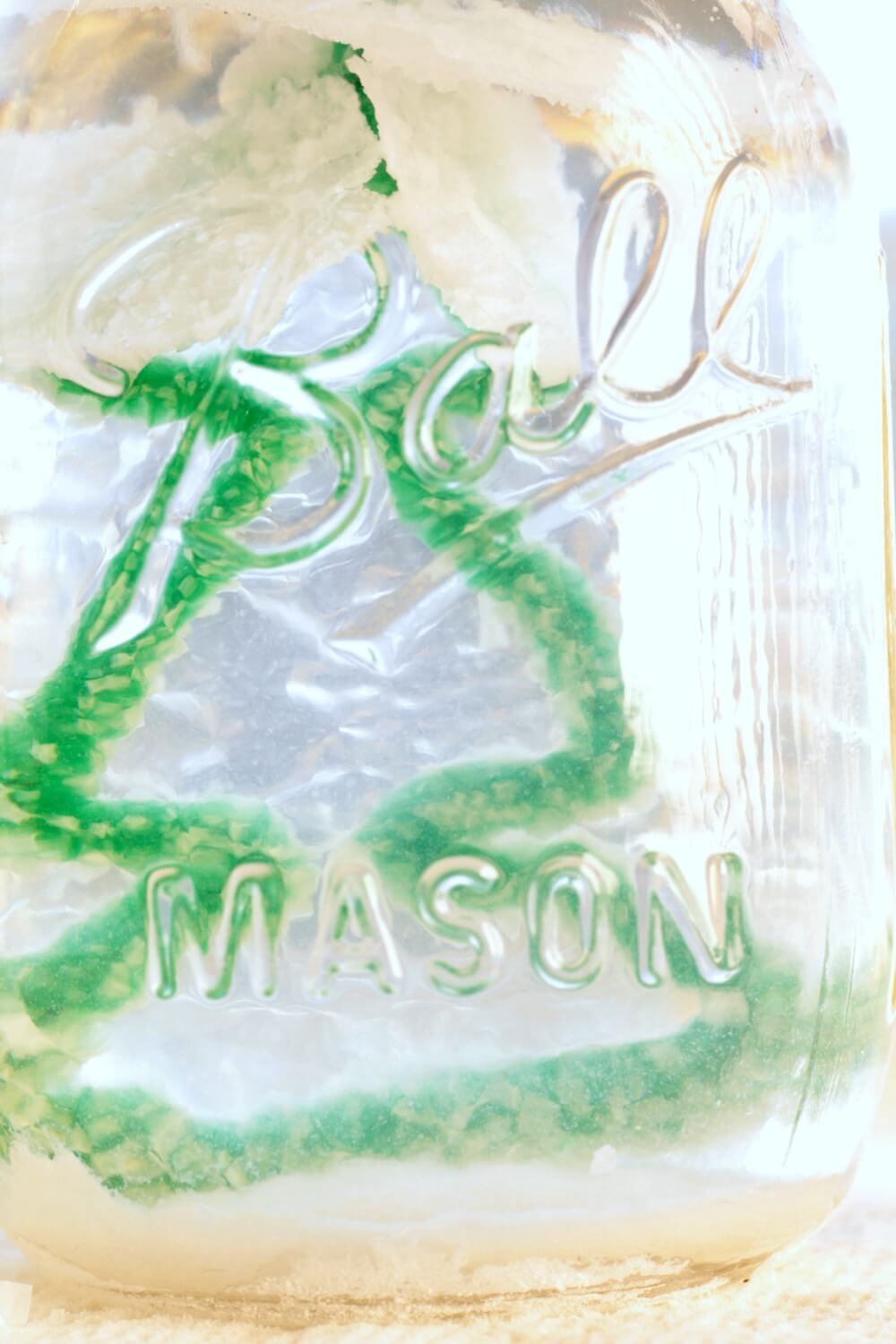
When the crystals look like this, they are ready to come out! The longer you wait, the more crystals will form. Try experimenting to see how different the ornaments look after one day in the salt solution, two days, etc.
Let your crystals dry completely.
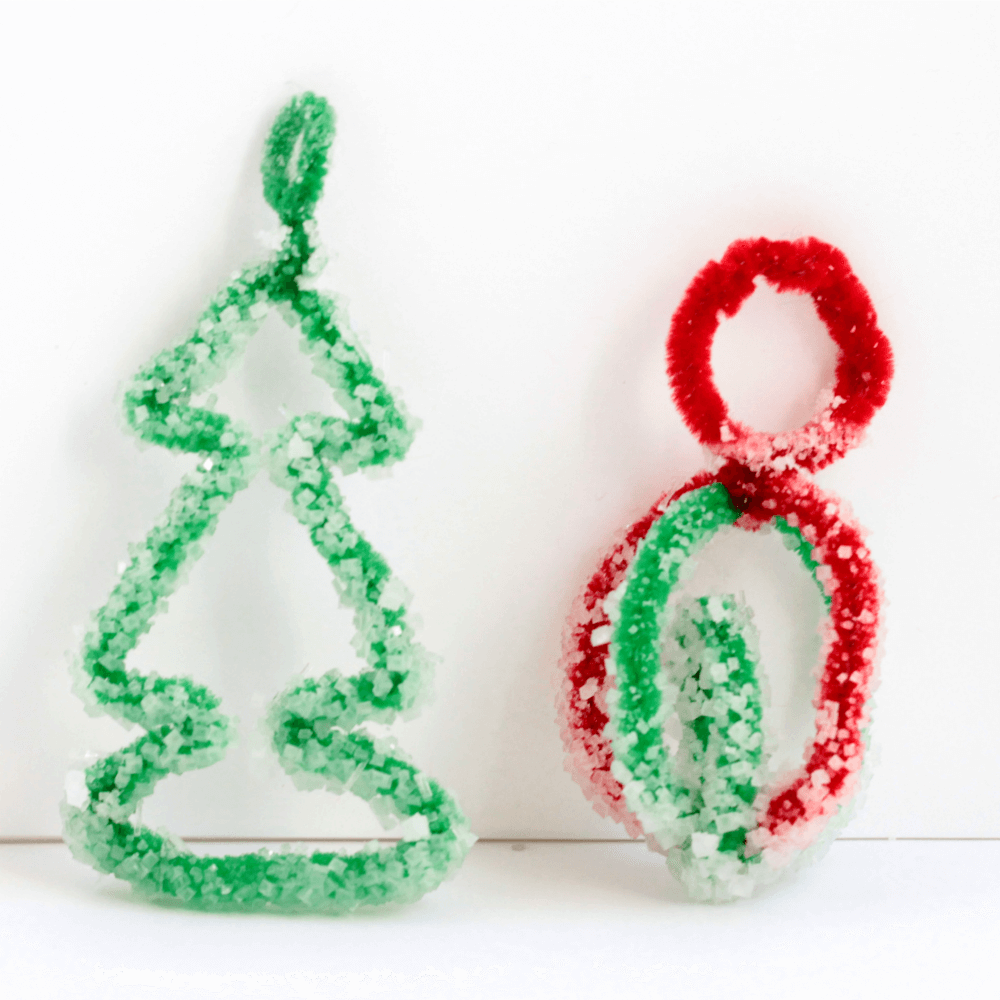
Add a chenille stem loop at the top of each ornament and they are ready to hang!
What Kids Learn by Making Salt Crystal Ornaments
The formation of crystals is all about ionic bonding. Salt crystals are formed in a specific pattern of ionic bonding which will always produce a square shape. When salt is dissolved into water, the water molecules separate the salt molecules. But as the water evaporates, the salt molecules bond once more. The crystals stick to the chenille stems because as the salt molecules fall to the bottom of the jar, some cling to the chenille stems, making the pretty crystal ornaments you can hang on your tree!

Looking for more STEAM (Science, Technology, Engineering, Arts and Math) projects and inspiration?
Check out my new book STEAM Kids Christmas and get a FREE STEAM Kids Coloring book if you buy during launch week! You can get both books (eBook PDF format) for only $9.99! But the sale and bonus book offer ends November 21, so grab your copy now and don’t miss out!

Get more information on STEAM Kids Christmas by clicking here.
Or go order your copy now! eBook PDF or Paperback

You can also get a bundle with our best selling STEAM Kids book, STEAM Christmas and STEAM Kids Coloring book for one low price: Click here to get your bundle today!
Not quite ready to jump in, don’t worry… you can hop over and grab a FREE Preview which includes 2 sample activities, 6 sample challenge cards, and the table of contents of the book!
For More Winter STEM Activities, Check Out:







I like the idea of making ornaments. I would want to get something that last long for my kids to see them. I would want to be able to have something unique on my tree.
Hi, im reading from Australia and have no idea what a regular sized tub of salt would be referring to. Can you please elaborate with weight or even dimensions of the tub? Thank you.